Plutonium in Space: Why and How?
Plutonium is a word that in some communities has very negative connotations. Plutonium was needed to create the atomic weapons of the Cold War, is highly regulated by proponents of nuclear nonproliferation, and is one of the causes of the environmental woes at the Hanford site in Washington State. On the other hand, it is also the element that has been used to safely power many space missions, including the Voyager, Galileo, Cassini, New Horizons, and the most recent Mars rover, Curiosity.
So, why is plutonium still used if it has issues associated with it?
The answer is that plutonium exists in multiple nuclear forms, or isotopes. Isotopes occur in elements naturally due to differing number of neutrons in the nucleus. While relatively unimportant on the chemical level, on the nuclear level isotopes of a single element can behave very differently. Plutonium-239, the isotope of plutonium with 94 protons and 145 neutrons, is a fissile isotope, meaning that after the absorption of a non-energetic neutron it has a possibility of splitting, or fissioning. Because of this capability, plutonium-239 can be used in nuclear reactors and weapons. Plutonium samples with a large fraction of the plutonium-239 isotope are referred to as weapons-grade plutonium.
However, devices that use plutonium to produce power use the plutonium-238 isotope, which has 94 protons and 144 neutrons. It is not fissile, and cannot be used in atomic bombs or nuclear reactors. Plutonium-238 is useful for radioisotope heat sources, and radioisotope power systems, because it decays radioactively, releasing a particularly useful form of radiation called alpha radiation.
Alpha radiation is simply energized and completely ionized helium atoms, which lose their energy in the form of heat when interacting with other matter. This energy loss mechanism is similar to how friction generates heat on a surface. Alpha radiation is generally not harmful to humans, provided its emitters are not inhaled or ingested; alpha particles can be stopped by the outermost layer of skin.
Pu-238 is safe and can produce heat, but why is it preferred over other power sources?
Radioisotope power systems are useful for space applications for two main reasons:
- First, they are very versatile. Unlike solar power sources, radioisotope power systems do not rely on correct orientation toward the sun, nor do they depend on proximity to the sun.
- Second, the power from plutonium-238 lasts a long time. The half-life of plutonium-238, or the amount of time it takes for the power produced by the isotope to decrease by half, is 87.7 years.
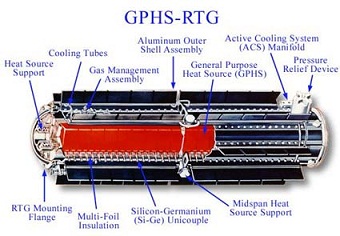
A power system fueled by plutonium-238 can last for a very long time. This is, of course, dependent upon the reliability of the heat-to-electricity conversion components. The most common power conversion method-a static system known as thermoelectric conversion-is very reliable and can last for decades.
Future radioisotope power systems will adopt a new method for power conversion called the Stirling cycle-a dynamic (moving) cycle-which will allow for higher efficiency and lower mass systems. The new generators will be termed Advanced Stirling Radioisotope Generators. For more information on radioisotope power systems, see this page maintained by the Department of Energy.
Where do we get plutonium-238? Can it be found naturally?
Unfortunately, plutonium-238 cannot be found naturally. This is because it is radioactive and will have almost completely decayed into a different element after a geologically short period of 1000 years. Thus, plutonium-238 must be produced using nuclear reactors.
During the Cold War, when weapons-grade plutonium production was at full scale, plutonium-238 was a byproduct that could be saved and used for space power production. Since the 1990s, however, the United States has stopped production of weapons-grade plutonium, yet we continue to plan space missions that require the use of plutonium-238. NASA and the DOE have discussed plans to use national laboratory reactors to produce plutonium-238 for general purpose applications, but it is questionable if they will be able to supply a sufficient amount to meet national needs.
Another concept, proposed by the Center for Space Nuclear Research (CSNR), uses flexible TRIGA research reactors to produce a higher quantity of Pu-238 per year at lower cost. For more information on low cost plutonium-238 production, contact the CSNR.
Regardless of its source, Pu-238 remains an important tool for scientific research. Many space missions have been powered by plutonium-238, and future missions will continue to be enabled by it. Its long lasting heat generation-coupled with a dependable power conversion system-allows it to be used in many environments and configurations. The use of plutonium-238 can be expected to become even more important as space exploration pushes further outward to Mars, Jupiter, their moons, and beyond!
This article is the first of a monthly series of ANS Nuclear Cafe entries on nuclear space topics by the ANS Aerospace Nuclear Science and Technology Professional Division.
_______________________________
Wes Deason is a graduate student in nuclear engineering at Oregon State University working on the safety analysis of vented fuel systems for gas-cooled fast breeder reactors. He is a former summer fellow for the Center for Space Nuclear Research and the current student liaison for the Aerospace Nuclear Science and Technology Division of ANS.